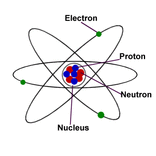
Intro
We get energy from the sun (solar panels) windmills and batteries. The chemistry of making batteries is called electro chemistry as we get older they will start making better and more efficient.
Oxidation Reduction: A reaction in which there is a transfer of electrons
Oxidation: the loss of electrons
Reduction: the gaining of electrons
- all elements have different attractions to elements
- When an atom that loses electrons easily than an atom of a different element come into contact with one another
Oxidation Reduction: A reaction in which there is a transfer of electrons
Oxidation: the loss of electrons
Reduction: the gaining of electrons
Looking back?
Looking back our experiment didn't turn out so well, I though we did everything right and I'm still not sure what we did wrong but our light didn't want to, light. There are images below showing, first using a sand paper to get rid of the copper on the penny. Then dipping the cardboard in vinegar, staking then over aluminum foil and finally adding the led which didn't even work.
looking beyond?
When CU and O come together copper becomes ozidized and oxygen becomes redoced.
Looking inward?
We definitely didn't get desirable results our LED didn't light up and we couldn't figure out what was wrong with it.
Looking outward?
For this lab I worked with Meech and Marco we all work very well and I don't think it was our partnership that messed up the project so I would work with them again
Looking forward?
If I could do this experiment again I would sand the pennies down even more so that there is absolutely no copper on one side. I see manny way in which we could continue this experiment we could try and light a larger light with more pennies and try to figure out the ratio of number of pennies:amount of watts you get.
Take aways?
My take away from this experiment was learning how to create energy out of cheap and easy to find materials