| | |
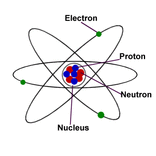
Today we did an experiment with magnesium, citric acid and water to learn about the structure of atoms. An atom is the smallest unit of any element. Atoms are made of subatomic particle such as protons (p+) neutrons (n^0) and electrons (e-). A proton (p+) gives off a positive charge is located in the nucleus and has a significant mass. An electron (e-) is a negative particle meaning it has a negative charge and has an insignificant mass. A neutron (n^0) has a neutral charge meaning it doesn't have a negative or positive charge. The neutron has a significant and is located in the nucleus.
Next we learned how an atom looks like as an element. Hydrogen was one of the first element found in space named by the sun. So its atomic number is one and It has an atomic mass of 1.0079. You can figure out the amount of protons and neutrons by the atomic number of the atom. Hydrogens atomic number is 1 so there is one proton and one neutron. To find out the number of neutrons there are in the atom you round the atomic mass to the nearest whole number and subtract it by its atomic number. If we were going to figure out the number of neutrons in hydrogen we would round 1.0079 to 1 and subtract its atomic number (1) and get 0.
Or experiment turned out very well and although we didn’t see the water we made we did get to see our balloon float and metal melt. Some of the observations I made were when all the ingredients were in the flask the flask started getting warm. I think the flask got warm because the atoms got close together and started shaking a lot and there's nowhere to escape so it started to get warm and the balloon got bigger.