Reflection
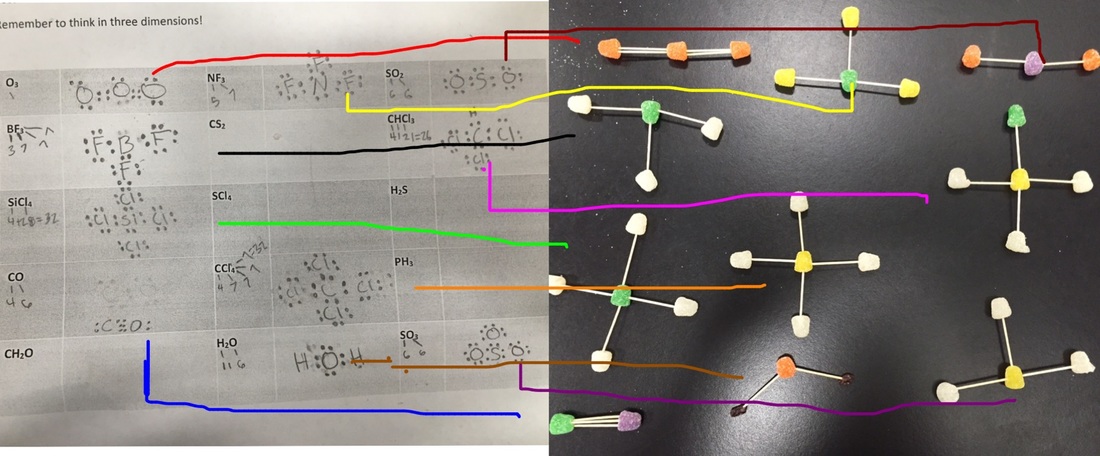
Today we did the molecule lab. This included building molecules from toothpicks, gumdrops and raisins. Although I didn't have raisins at my house I just used a different color gumdrop like the color red when I was making the aspirin and I used yellow when I was making caffeine and nicotine. We also leaned about the organic compounds and bonds.
"Ionic bonds are between a non-metal and a metal and
occur when charged non-metal atoms (or ions) attract
electrons from a metal
This occurs when a metal ion loses one or more electron to
the non metal
Example: NaCl
Covalent Bonding are bonds are between two non-
metal atoms. Both atoms need to gain electrons, so
the electrons are shared
This occurs when both non-metal atoms lack a full outer
shell of eight electrons
Example: H2O"(Amandas blog)
"Ionic bonds are between a non-metal and a metal and
occur when charged non-metal atoms (or ions) attract
electrons from a metal
This occurs when a metal ion loses one or more electron to
the non metal
Example: NaCl
Covalent Bonding are bonds are between two non-
metal atoms. Both atoms need to gain electrons, so
the electrons are shared
This occurs when both non-metal atoms lack a full outer
shell of eight electrons
Example: H2O"(Amandas blog)